Products and services
Testing Services


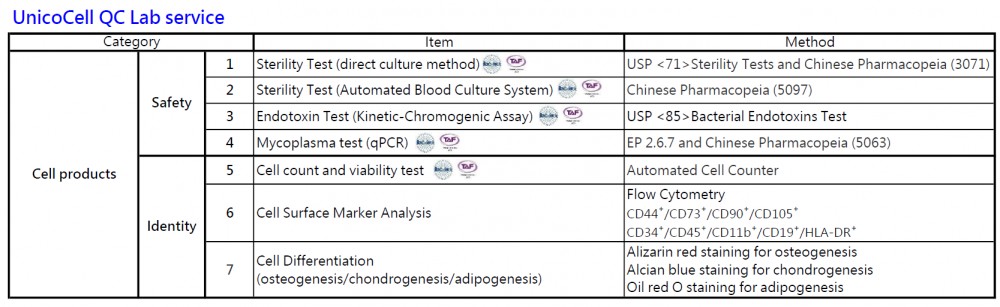
UnicoCell's quality control laboratory has obtained ISO/IEC 17025 accrediation granted by Taiwan Accreditation Foundation (TAF).
We offer the following testing services: "sterility test", "endotoxin", "mycoplasma", "cell count and viability".
Product Description

We offer testing services for safety and identity test listed below. For more information, please contact info@unicocell.com